Here is a term paper on ‘Plant Tissue Culture’. Find paragraphs, long and short term papers on ‘Plant Tissue Culture’ especially written for school and college students.
Term Paper on Plant Tissue Culture
Term Paper Contents:
- Term Paper on the Introduction to Plant Tissue Culture
- Term Paper on the Meaning of Plant Tissue Culture
- Term Paper on the Types of Plant Tissue Culture
- Term Paper on the Applications of Plant Tissue Culture
Term Paper # 1. Introduction to Plant Tissue Culture:
In 1902, a German botanist Gottlieb Haberlandt developed the technique to culture the isolated cells of Tradescantia in artificial conditions. But he could not induce cell division. In 1904, Hannig isolated embryos of some crucifers and grew it on sugar and salt solution. In 1908, Simon successfully regenerated a bulky callus, buds, and roots.
In 1939, P.R. White an American scientist, reported for the first time the successful continuous cultures of tomato root tips in liquid medium and in-vitro cultivation of viruses on excised roots. F. Skoog and C.O.
Miller in 1959 discovered kinetin and demonstrated induction of regeneration of shoots in tobacco callus. This was a landmark in multiplication of plants by tissue culture. G.M. Morel laid the foundation of commercial plant tissue culture in 1960 for a million fold increase in clonal multiplication of orchid.
Depending on the type of in-vitro growth plant tissue culture can be classified into two categories – callus and suspension culture. Plant tissue culture can also be classified on the basis of explants used for culturing – embryo culture, anther culture, etc.
Explant is that organ or tissue of plant, which is excised and used for in-vitro culture. Depending on the purpose of culture, any part of the plant can be used as an explant. Before starting the culturing, it is mandatory that each and every equipment is sterilised and the surrounding is totally aseptic. This not only includes all the instruments but also the explant and the growing medium.
For making everything free from microbes they are treated with anti-microbial chemicals. They can also be made aseptic by treating with alcohol, steam (autoclave), dry heat, etc. This is known as surface sterilisation. Explants are generally treated by 1-2% solution of sodium or calcium hypochlorite or with 0.1% solution of mercuric chloride.
Composition of culture medium governs the growth and morphogenesis of plant tissue in-vitro. Generally, cultured tissue requires the same nutrients as the plant. But cultures grown in the laboratory require some special components that promote maximum growth of the tissue under laboratory conditions. Composition of culture medium varies with the type of plant cell or tissue used for culture.
The principal constituents of plant tissue culture medium are carbon sources, inorganic nutrients, organic supplements, growth regulators and gelling agents. Carbon and energy source is mostly sucrose and glucose. Once sucrose is autoclaved it changes to glucose and fructose. These help in proliferation of cells and tissue regeneration. Inorganic nutrients are nitrogen, phosphorus, potassium, magnesium, calcium and sulphur- macronutrients.
Some essential micro-nutrients are copper, iron, zinc, manganese, cobalt, boron and molybdenum. Organic supplements are required for growth of the tissues. They are vitamins, amino acids (arginine, glycine, asparagine, glutamine, cysteine), coconut milk, yeast and malt extract. Ground banana, orange juice and tomato juice can also be used. If needed antibiotics like streptomycin can also be added to culture medium to avoid infection of micro-organisms. Growth regulators are required for proliferation of cultured tissue.
Auxin (IAA, NAA, 2, 4 – D: chlorophenoxyacetic acid) induces cell division and causes elongation of internode and stem. Cytokines (kinetin benzylaminopurine) induce cell division and differentiation of shoot. More of auxin promotes embryogenesis, root and callus initiation, whereas more of auxin leads to shoot and axillary promotion. Agar is generally used as Gelling agent. Optimum pH of a culture is between 5.0 and 6.0. Optimum temperature is around 24°C with some illumination.
Term Paper # 2. Meaning of Plant Tissue Culture:
Plant tissue culture means to cultivate the plant cells, tissues, organs or a complete plant in aseptic conditions in a suitable culture medium in-vitro. The scientific process by which cells, tissues or organs are developed in nutrient media to fulfil the demand, is called plant tissue culture.
The plant part taken out to be grown in a test-tube in special nutrient media is called explant. And the capacity of producing a whole plant from explant is called totipotency. The nutrient media must provide a carbon source (such as sucrose), inorganic salts, vitamins, amino acids and growth regulators such as auxins, cytokinins, etc.
The production of a large number of plants in a short duration is practiced in floriculture and horticulture industry today, is known as micro-propagation. It is a technique of obtaining large number of plantlets by the tissue culture method.
The plants thus, generated are genetically similar to the original plant through which they are grown, so they are called somaclones. To recover healthy plants from diseased plants, meristem culture is practiced. Meristem (axillary and apical) is the only virus-free part of virus-unaffected plant. Plants developed from meristem are banana, sugarcane, potato, etc.
Term Paper # 3. Types of Tissue Culture:
a. Meristem Culture:
In this kind of culture, the shoot apical meristem is used. This is used for quick vegetative propagation and is used extensively. Explant used for this method are shoot tip and nodal segment. In this, the shoot tip can be cut into many pieces to get more than one plant. During meristem culture the viruses can be eliminated to get virus free plants. The plantlets obtained are subjected to hardening and then established in the fields. Meristem culture is used for germplasm conservation and production of transgenic plants (Fig. 10).
b. Embryo Culture:
This method involves excision of young embryo from developing seeds and cultivating them on a nutrient medium. In some cases like orchids, embryo of mature seed can also be used for culture. The main objective of this is to allow the young embryos to complete development and ultimately give rise to seedlings.
Following are the applications of this method:
i. The plants in which seeds lack stored food; their embryos can be cultured by this technique.
ii. In some seeds endosperm has inhibitors, which make the seeds dormant. In such cases, embryo culture helps in overcoming this problem.
iii. In certain interspecific crosses there is degeneration of endosperm of developing seed at an early stage. This results in the subsequent degeneration of growing embryo. In such cases the embryo is excised and cultured in-vitro. This technique is also known as embryo rescue.
c. Anther Culture and Haploid Production:
Anther culture means producing plantlets by culturing anthers. This gives rise to haploid plants (Fig. 11). In India, this was used to produce Datura which is haploid. The stage at which the pollen is to be cultivated is different for different species e.g. in Nicotiana sp., the pollens are cultured when they are in binucleate stage whereas in cereals they should be in uninucleate stage.
Continuous division of pollen gives rise to a callus, which later generates the shoot and then root. In some cases, ovaries or ovules can also be used to produce haploid plants. Haploids produced by the above method are sterile but they can be used to produce homozygous lines. For breeding program pollens from F1 plant are cultured to obtain haploid plant, which is later made diploid by using colchicines. Now these plants are homozygous. Now the plants breeders can select the plants of desired characters.
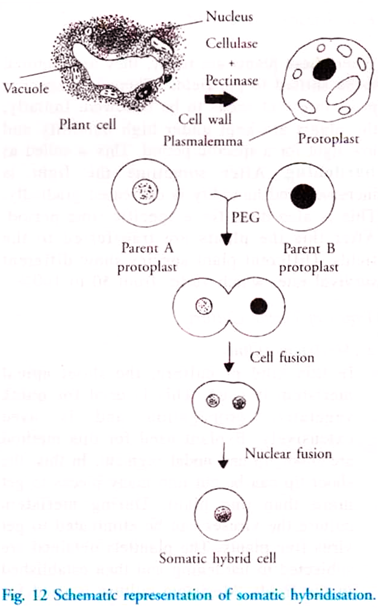
d. Protoplast Culture and Somatic Hybridisation:
Fusion of somatic cells of different parents to produce a hybrid is called somatic hybridisation (Fig. 12). Plants produced by this method are called somatic hybrids.
This process follows the following steps:
i. Isolation of Protoplast:
The first step of hybridisation is the removal of cell wall. This is done with the help of enzyme pectinase and cellulose. These two enzymes help in digesting the cell wall. The cells without the cell wall are called protoplast.
ii. Fusion of Protoplast:
This is induced by polyethylene glycol (PEG) or by very high voltage current.
iii. Formation of Hybrid Cell.
iv. Regeneration of a Hybrid Plantlet:
Somatic hybridisation helps in production of hybrids which cannot be produced by means of sexual hybridisation. This technique is used in production of hybrids, which is the fusion of protoplast of one species with the cytoplasm of the other. Such varieties show herbicidal resistance gene. Bromato and pomato are two hybrids produced by this technique. Bromato is hybrid between brinjal and tomato and pomato is hybrid between potato and tomato.
Somatic hybridisation also helps in producing hybrids between non-flowering angiospermic plants.
Term Paper # 4. Application of Tissue Culture:
Plant tissue culture is a useful technique in formation of new plantlets. Generation of plantlets by this technique is known as plant biotechnology.
Following are a few applications of tissue culture:
a. Production of virus resistant plants by meristem culture
b. Production of interspecific hybrids
c. Rapid clonal propagation
d. Shortening of breeding cycle
e. Somatic hybrids by protoplast fusion
f. Creation of transgenic plants. Transgene is the gene which is transferred into an organism by genetic engineering. Organism containing and expressing this gene is called transgenic organism
g. Embryo rescue for viable hybrids
h. Haploid plants by anther culture.


No comments yet.