The following points highlight the two main principles of plant tissue culture. The principles are: 1. Callus Culture 2. Organ Culture.
Principle # 1. Callus Culture:
For raising the callus tissues, a tissue culturist must have clear understanding of basic principles. All types of organs like shoot apex, bud, leaf, mesophyll cells, epidermis, cambium, anthers, pollen, fruit etc., when inoculated on a suitable medium under aseptic laboratory conditions are able to differentiate and multiply – callus is induced.
This results into the formation of an amorphous mass of cells known as callus, which can be induced to re-differentiate on appropriate medium to develop embryoids which directly develop into plantlets, eventually giving rise to a whole viable plant. Whereas in nature wound and pathogens induce callus tissue formation in intact plants, in tissue culture, callus formation is artificially induced in explants grown in micro-environments.
The term clone (derived from Greek work klon), meaning a twig suitable for plant propagation) was suggested by Webber (USA) to explain those plants which were obtained by a sexual reproduction. Also means an organism obtained from a single cell through mitotic divisions. Formation of callus is the result of expansion and cell division of cells of explants.
Calluses can vary in appearance and texture; generally yellow or white in colour, or often pigmented. Secretion of phenolic compounds gives brown colour to the callus. By doing repeated sub-culturing of callus, it is able to grow on a culture medium which is devoid of growth hormones.
An exogenous supply of regulators is often recommended to induce callus on an explant:
(i) Auxin only required (especially monocotyledons).
(ii) Cytokinin alone needed.
(iii) Both auxin and cytokinin required.
Besides growth regulators, genotype, glucose (2 – 4%), coconut milk, light, nutrient composition, high temperature (22 – 28°C) govern callus formation.
Principle # 2. Organ Culture:
A particular organ is isolated and cultured under laboratory conditions in a defined medium where they retain their characteristic structures and other features and continue to grow as usual. In organ culture, organs are not induced to form callus, therefore, it differs from the callus culture where the organization of the intact tissues is lost. This technique provides an. experimental system to define the nutrients and growth factors that are usually received by the organ from other parts of the plant body and from surrounding environment.
Thus, organ culture offers an excellent experimental system to study:
(i) Nutritional requirements of the organ (explant).
(ii) Environmental effects.
(iii) Metabolite biosynthesis.
(iv) Development of new varieties.
a. Root Culture:
It refers to culture of excised radicles of aseptically germinated seeds. Root has been cultured successfully and induced to regenerate under in vitro conditions. Genetic stability associated with growing tips offers roots as a suitable material for large-scale micro-propagation of desired plants. There are appreciable amounts of successful reports of root culture (Table 1.1).
Root culture, besides offering a good material for micro propagation, is also used for production of pharmaceutically important secondary metabolites of high commercial value in case of hairy root culture). This method also enables us to understand the process of growth and differentiation in roots besides the physiological requirements of these processes.
b. Stem Culture:
The stem segments have been the most frequent explant used for in vitro studies (Table 1.2). These explants could be induced for direct or indirect regeneration. The callus cultures obtained from stem segment have often been looked upon as a source of soma clonal variations and a rich nutrient for secondary metabolite analysis. The ultimate success with stem segment culture depends upon the media composition, physiological status of the explant and the in vitro environment.
c. Shoot Tip/Meristem Culture:
Shoot tips have got apical meristem and thus possess inexhaustible degree of regeneration potential. It refers to culture of terminal (about 0.1 – 1.0 mm) part of shoot having meristem together with primordial and developing leaves and adjoining stem tissue also called as ‘mericloning’. Clonal propagation of woody plants, ornamentals and rare species has been possible through this method (Table 1.3). It is useful in developing virus – free plants and micro propagation and storage of genetic resources. It also provides a suitable material for biochemical, physiological and genetical studies.
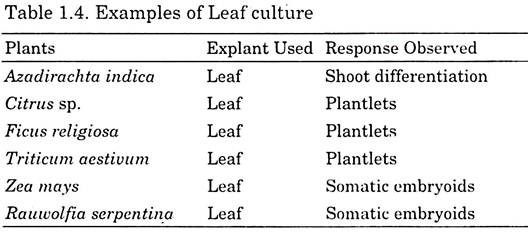
In leaf, only a few layers of cells can respond to in vitro conditions and give a great degree of totipotency. The apical cells get activated and undergo morphogenic changes to form a plant. Leaf cultures have also been used for callus induction and secondary metabolite synthesis. Here, excised young leaf primordia are cultured (Table 1.4).
e. Flower Culture:
Culture of flower primordia or flower bud is the basis for flower culture. Flower culture is useful in studies concerned with flower and fruit development, sex differentiation etc., e.g. Arachis hypogea, Allium cepa, Tagetes sp.
f. Ovary Culture:
It refers to culture of ovaries excised from pollinated or non-pollinated flowers. It is useful in obtaining hybrids between self-incompatible species and induction of polyembryony. Through ovule culture it is possible to obtain seedlings from crosses that are unsuccessful.
g. Anther and Pollen Culture:
This method is useful in developing haploid plants to get homozygous diploids. This technique finds its role in plant breeding to maintain male sterile lines.





No comments yet.