In this article we will discuss about:- 1. Growth Profile of Plant Tissue Culture 2. Growth Measurement of Plant Tissue 3. Methods 4. Types.
Growth Profile of Plant Tissue Culture:
The growth profile of the plant tissue culture can be classified under two headings: 1. Single Cell Culture 2. Callus Culture.
1. Single Cell Culture:
The single cell culture exhibits various stages of growth (Fig. 6.2).
i. Lug Phase:
Here, the cells are exposed to a fresh medium; the cell starts or regains the ability to divide and the tissue shows slow growth.
ii. Exponential Phase:
This phase is characterised by rapid cell multiplication. The time of this phase varies according to the cell and its nutrient regime. Usually, it is of short duration and lasts for 3-4 generations.
iii. Linear Phase:
In this stage, the growth follows a linear pattern with respect to time.
iv. Progressive Deceleration Phase:
Here, the rate of cell division decreases with the aging of the culture.
v. Stationary Phase:
In this stage no growth of cells occurs. The rate of production of cells is equal to the rate of their death.
vi. Senescent Phase:
Here, the cells die due to unavailability of nutrients.
2. Callus Culture:
The various stages of the growth profile of a callus culture (Fig. 6.3) are as follows:
i. Lag Phase:
After inoculation of the explant into the nutrient media, there is a lag time in which the cells undergo no growth; this is when the cell is trying to adjust to the new nutritional environmental conditions.
ii. Exponential Phase:
Here, the cells undergo rapid multiplication and consume nutrients from the medium leading to their depletion.
iii. Decline Phase:
Depletion of the nutrient elements in the media lead to the starvation of some cells which in turn leads to the decline in the growth of callus tissue.
iv. Stationary Phase:
From this stage onwards, no growth is evident. Sub-culturing of the cells is required for further growth and development.
Growth Measurement of Plant Tissue:
There are various techniques available for measuring culture growth of plants: 1. Cell Number 2. Packed Cell Volume 3. Fresh and Dry Weights 4. Nutrient Uptake Studies 5. Cell Viability Measurements.
1. Cell Number:
This is a simple method which involves the direct counting of the cells. It gives accurate information about the number of cells. The cells are treated with pectinase or chromic trioxide, and then counted directly.
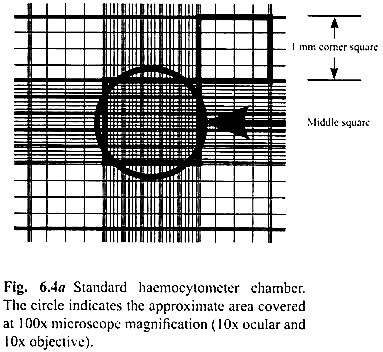
Determination of the total cell-count by using a haemocytometer is described below:
a. Prepare a cell suspension in a balanced salt solution containing approximately 2.5 × 105 cells per ml.
b. With the cover-slip in place, use a Pasteur pipette or any other suitable device to transfer a small amount of cell suspension to both chambers of the haemocytometer by carefully touching the edge of the cover-slip with the pipette tip and allowing each chamber to fill by capillary action. Do not overfill or under-fill the chambers.
c. Starting with one chamber of the haemocytometer, count all the cells in the 1 mm centre square and four 1 mm corner squares (Fig. 6.4a).
Note:
Count cells on the top and left touching the middle line of the perimeter of each square. Do not count cells touching the middle line at the bottom and right sides (Fig. 6Ab).
d. Repeat this procedure for the second chamber.
Note:
If more than 10% of the cells appear clustered, repeat the entire procedure making sure that the cells are dispersed by vigorous pipetting in the original cell suspension. If less than 200 or greater than 500 cells (20-50 cells per square) are observed in the 10 squares, repeat the procedure adjusting to an appropriate dilution factor.
e. Cell counts each square of the haemocytometer, with the cover-slip in place, represents a total volume of 0.1 mm3 or 10–4 cm3.
Since 1 cm3 is equivalent to 1 ml, the subsequent cell concentration per ml (and the total number of cells) will be determined using the following calculations:
Cells per ml = the average count per square × dilution factor × 104 (count 10 squares)
Example:
If the average count per square is 45 cells, the cells per ml is
45 × 104 = 4.5 × 105 cells/ml
Total cells = cells per ml × the original volume of fluid from which cell sample was removed
Example:
4.5 × 105 (cells per ml) × 10 ml (original volume) = 4.5 × 106 total cells.
f. Withdraw a second sample and repeat the counting procedure to ensure accuracy.
2. Packed Cell Volume:
Here, a known volume of the suspension culture is subjected to centrifugation and the cells allowed to settle at the bottom. Then, the total volume occupied by the cells is calculated as a percentage of the total volume of suspension. This method only gives approximate results.
3. Fresh and Dry Weights:
This is the most widely used method of measuring culture growth. In the linear phase, the cells prepare cell wall material and starch from the available carbohydrates. At the progressive deceleration and stationary phases, the available carbohydrates are metabolised by the cells.
Automatically, the synthesis of starch and its accumulation is stopped. Hence, there is a decline in the dry weight corresponding to the metabolism of accumulated starch. But there is also a progressive increase in fresh weight as the cells are larger and trap culture medium on the filter bed.
4. Nutrient Uptake Studies:
Here, the growth measurement is carried out by analysing a certain specific nutrient element in the media like glucose, sucrose, inorganic phosphates, etc., which indicates if a particular component is limiting at any stage in the culture cycle. From the date of carbohydrate analysis, the carbon conversion efficiency is calculated and the biomass subsequently measured.
5. Cell Viability Measurements:
The ability of a cell to live and grow is called cell viability. Depending upon the metabolic functions, the following viability tests can be performed.
a. Cytoplasmic Streaming:
This method is applicable only to cell suspensions, protoplasts and single clumps of cells through which light can pass. It is a non-destructive method of measuring the growth profile of the cells.
b. Measuring Membrane Integrity:
This method can be applied to all types of cultured plant materials. Different types of cells in tissues leak electrolytes at different rates. This electrolyte loss can be measured from cells having contact with water.
c. Measuring Biochemical Activity:
It involves the use of 2, 3, 5-triphenyl tetrazolium chloride (TTC). In the presence of TTC solution, living cells turn red whereas the dead tissue remains unchanged. The red complex precipitate is extracted from the cells with alcohol and absorbance is measured spectrophotometrically.
Methods of Plant Tissue Culture:
The following methods of tissue culture are in practice:
1. Hanging Drop Culture:
This is the simplest method. It involves the following steps:
a. A tissue of approximately 1 mm diameter is taken and placed on a cover-slip containing a drop of solidified medium.
b. A grooved slide of 1″ × 3″ size is taken and the cover-slip is placed over it.
c. The edges of the cover-slip (rim region) are sealed with wax.
In this type of culture, the viability of the tissue is very poor. The tissue can be made viable for a longer period of time by frequently changing the culture medium i.e., by the sub-culturing method. In sub-culturing, the tissue is divided into smaller parts and each part is sub-cultured in a new grooved slide with fresh nutrient medium.
2. Double Cover-Slip:
This method was developed by Torrey and Maximov for culturing pea root callus on solidified medium. Since it involves the use of two cover-slips on a grooved slide, it is termed as the double cover-slip method.
It involves the following steps:
a. A small round cover-slip containing a tissue and the nutrient medium is placed on a larger square cover-slip.
b. The small cover-slip is attached to the larger cover-slip by a drop of water or salt solution and the edges of the cover-slip are sealed with wax.
c. The culture medium must be changed frequently. For this, the small cover-slip is detached from the larger cover-slip and washed with a fresh nutrient solution. Again the
cover-slip is attached to the larger cover-slip, placed in a new grooved slide with the edges sealed.
3. Culture in Steward’s Auxophyton:
This method was developed by Steward, Caplin and Miller, for the proliferation of small explants from carrot root. The culture apparatus used for the purpose is called auxophyton.
It involves the following steps:
a. Select a tube of 12.5 cm length and 3.5 cm diameter in size having a side neck at the middle.
b. Introduce the medium (10 ml) and the explant into the tube, closed with a cotton plug, which helps in gaseous exchange.
c. About 24 such tubes can be mounted along the edge of a disc which is rotated at a slight angle to the horizontal at a speed of 1-2 revolutions per minute. It allows the tissue to be exposed to air and the liquid medium, which flows along the tube from end to end alternately.
The apparatus is kept in a temperature controlled room with a fluorescent light. The culture tubes are uniformly illuminated. The medium generally becomes turbid due to the release of free cells and cell aggregates from the explants. The suspension itself may be used for the sub-culture.
4. Culture in Bottles, Conical Flasks:
Here, conical flasks, bottles or Petri dishes are used as culture vessels.
It involves the following steps:
a. A culture vessel of suitable size is selected and nutrient medium is poured into it.
b. The tissue is inoculated on the media. In due course of time, the cells undergo proliferation on the surface of the bottle or conical flask, forming a monolayer.
c. Frequent sub-culturing or changing the medium is necessary to maintain the viability of the cells for a longer period of time.
5. Culture in Microchamber:
Jone, Hildebrandt and Riker cultured the cells from a callus of the hybrid Nicotiana tabcum and Nicotiana glutinosa in a microchamber which was sealed with inert mineral oil. The cells can be minutely observed in such a culture.
6. Culture in Petri Dish, Watch Glass:
Organs may be cultured in a Petri dish or watch glass. The Petri dish may be incubated in a humidified incubator. For culturing small number of cells, the Petri dish is recommended.
Types of Plant Tissue Culture:
Tissue culture can be categorised into the following types:
Static Culture:
In static culture, cell proliferation takes place on solidified nutrient agar medium. It is also called callus culture.
Suspension Culture:
It is the culture of cells or cell aggregates dispersed in a moving liquid medium. Cells of a suspension culture often possess large nuclei, dense cytoplasm and starch grains. They show hormone habituation, increase in ploidy level and loss in totipotency. But cell suspensions from different sources vary considerably in the expression of these characteristics.
Subculture:
When the culture has reached the maximum cell density, sub-culturing should be done. Otherwise, the cells may die and lyses occur. Sub-culturing is usually done at the early stationary phase or during the progressive deceleration phase. In a suspension culture, maximum cell density is usually reached within 18-25 days. But in some cases, maximum cell density is reached within 6-9 days.
Before the first subculture, the culture is passed through a stainless steel filter or nylon to remove cell aggregates and residual in culture. By repeated subculture, a cell suspension culture can be maintained for a long time.
Batch Suspension Culture:
The cells are grown in a fixed volume of medium. It is a closed system of culture.
Continuous Suspension Culture:
A constant flow of the culture medium is maintained in the culture vessel, that is, there is a replacement of equal volume of culture medium after depletion. New cell formation is more or less equal to the amount of old washed out cells. Thus, the culture is maintained in a steady condition.
In an open system, fresh medium is added, the spent medium is withdrawn and the cells are also harvested.
In a closed culture, all cells remain in the culture, thus the number of cells increases until the stationary phase is reached. Carrot culture is usually done by this method.
Significance of Suspension Culture:
It is possible to synthesise secondary plant products such as alkaloids, glycosides, etc., by suspension culture. It also solves many problems in applied botany.




No comments yet.